All content on this site is intended for healthcare professionals only. By acknowledging this message and accessing the information on this website you are confirming that you are a healthcare professional. If you are a patient or carer, please visit the MPN Advocates Network.
The MPN Hub website uses a third-party service provided by Google that dynamically translates web content. Translations are machine generated, so may not be an exact or complete translation, and the MPN Hub cannot guarantee the accuracy of translated content. The MPN Hub and its employees will not be liable for any direct, indirect, or consequential damages (even if foreseeable) resulting from use of the Google Translate feature. For further support with Google Translate, visit Google Translate Help.
The MPN Hub is an independent medical education platform, sponsored by AOP Health, GSK, and Novartis, and supported through independent educational grants from Blueprint Medicines, Incyte, Bristol Myers Squibb and Merck Sharp & Dohme LLC, Rahway, NJ, USA. The funders are allowed no direct influence on our content. The levels of sponsorship listed are reflective of the amount of funding given. View funders.
Now you can support HCPs in making informed decisions for their patients
Your contribution helps us continuously deliver expertly curated content to HCPs worldwide. You will also have the opportunity to make a content suggestion for consideration and receive updates on the impact contributions are making to our content.
Find out more
Create an account to access:
Bookmark & personalize site content
Receive alerts for new content in your areas of interest
View MPN content recommended for you
International Consensus Classification 2022 for myeloproliferative neoplasms
There has been major progress in disease understanding and management of myeloproliferative neoplasms (MPN) with new clinical, morphological, and genomic data. These advances have prompted updates to available guidelines for diagnosis criteria. For example, the World Health Organization (WHO) Classification of Haematolymphoid Tumours has recently been updated and we summarized the key updates for MPN here.
In addition, a new International Consensus Classification (ICC) has been introduced for myeloid neoplasms and acute leukemia, as a separate system, by experts involved in prior editions of the WHO classification.1 In the context of MPN, the classical subtypes of MPN remained unchanged; however, the group made an effort to refine the diagnostic criteria to allow a distinction between subtypes. Below, we summarize the diagnostic criteria, along with the key points from the new ICC framework.
BCR-ABL1-negative MPN subtypes1
Essential thrombocythemia, polycythemia vera, and primary myelofibrosis are grouped under the category of BCR-ABL1-negative MPN subtypes, and the new classification aimed to increase diagnosis certainty in early-stage disease. Guidance on diagnosis is largely unchanged, and the combination of molecular data with bone marrow morphology and blood counts remain the current recommended diagnostic markers. The full diagnostic criteria for the common BCR-ABL1-negative MPN subtypes are shown in Figure 1.
Figure 1. Diagnostic criteria for common BCR-ABL1-negative MPN subtypes*
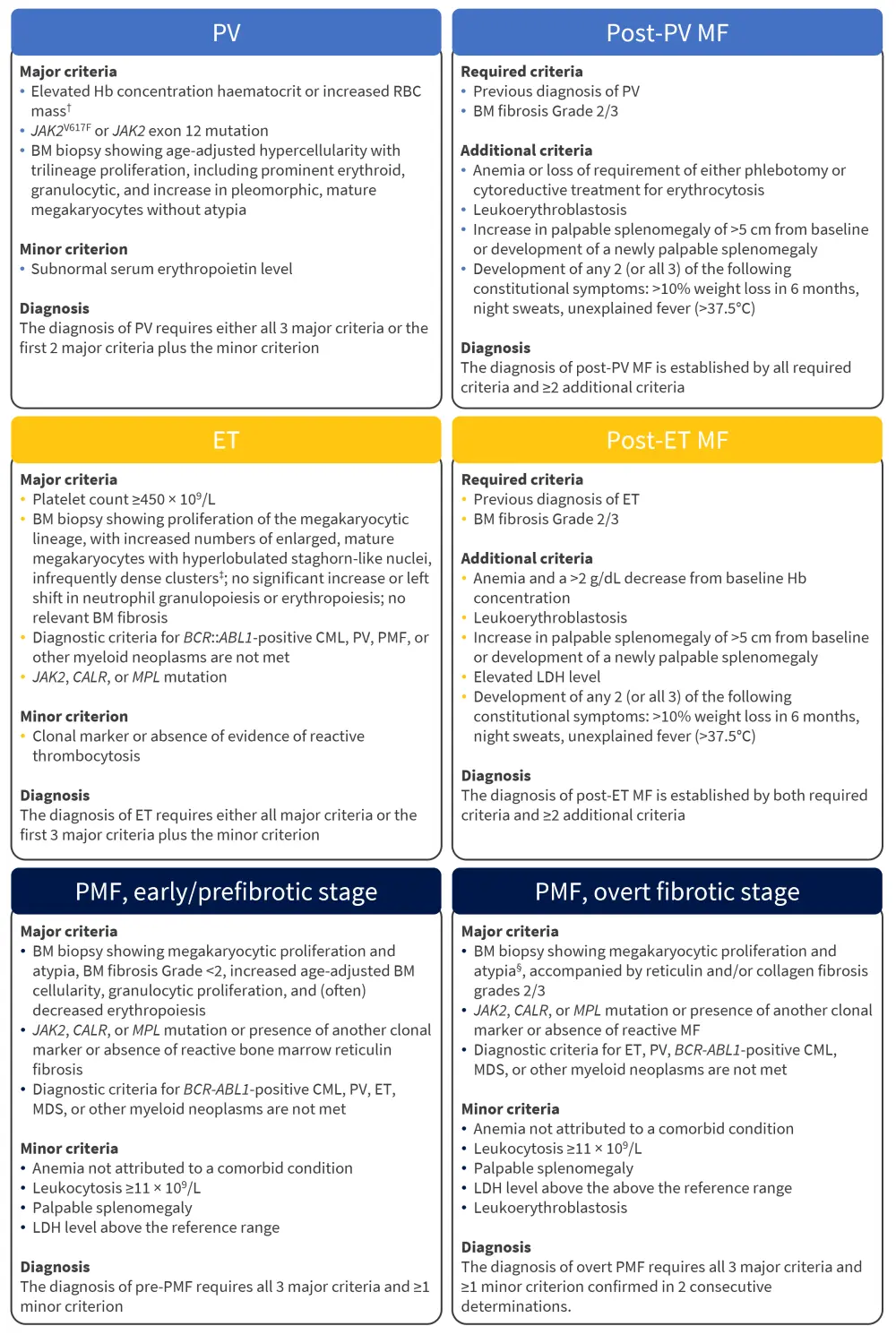
BM, bone marrow; CML, chronic myeloid leukemia; ET, essential thrombocythemia; Hb, hemoglobin; LDH, lactate dehydrogenase; MDS, myelodysplastic syndrome; MF, myelofibrosis; PMF, primary myelofibrosis; PV, polycythemia vera; RBC, red blood cell; WBC, white blood cell.
*Adapted from Arber, et al.1
†Diagnostic thresholds: Hb >16.5 g/dL in men and >16.0 g/dL in women; hematocrit >49% in men and >48% in women; RBC mass > 25% above mean normal predicted value.
‡≥3 megakaryocytes lying adjacent without BM cells in between; in most of these rare clusters ≤6 megakaryocytes may be observed, increase in huge clusters (>6 cells) accompanied by granulocytic proliferation is a morphological hallmark of pre-PMF.
§Morphology of megakaryocytes in pre-PMF and overt PMF usually shows a higher degree of megakaryocytic atypia than in other MPN subtypes; features of megakaryocytes include small to giant megakaryocytes with a prevalence of severe maturation defects (cloud-like, hypolobulated, and hyperchromatic nuclei) and presence of abnormal large dense clusters (mostly >6 megakaryocytes lying strictly adjacent).
Other MPN subtypes1
Chronic neutrophilic leukemia
Chronic neutrophilic leukemia, a notably rarer BCR-ABL1-negative subtype, is commonly identified by driver mutations in CSF3R. However, the absence of mutations in CSF3R does not exclude the possibility of chronic neutrophilic leukemia, as abnormalities in SETBP1, ASXL1, and SRSF2 are also feasible and indicate a potential diagnosis. The new classification has suggested reducing the diagnostic threshold of leukocytosis from ≥25 × 109/L to ≥13 × 109/L in patients with CSF3RT618I or CSF3R mutations. The full diagnostic criteria for chronic neutrophilic leukemia are shown in Figure 2.
Chronic eosinophilic leukemia
Chronic eosinophilic leukemia is an MPN identified by eosinophilia but falls outside of the criteria used to describe other defined subtypes. The new ICC includes the addition of abnormal bone marrow histopathology into the current diagnostic criteria to help confirm the neoplastic nature of the subtype, as well as distinguishing from the related entities, idiopathic hypereosinophilic syndrome and hypereosinophilic syndromes of unknown significance. The full diagnostic criteria for both chronic eosinophilic leukemia and idiopathic hypereosinophilic syndrome are shown in Figure 2.
MPN, unclassifiable
The category of MPN, unclassifiable is reserved for patients who present with clinical characteristics that lack important diagnostic features (Figure 2) that would normally allow for a clear diagnosis of another subtype. It also encompasses patients presenting with very early phase disease who require close monitoring for the development of specific subtypes that may only be detectable at follow up. In addition, MPN, unclassifiable includes patients with splanchnic or portal vein thrombosis who do not meet diagnostic criteria listed for other subtypes.
Figure 2. Diagnostic criteria for other MPN subtypes*
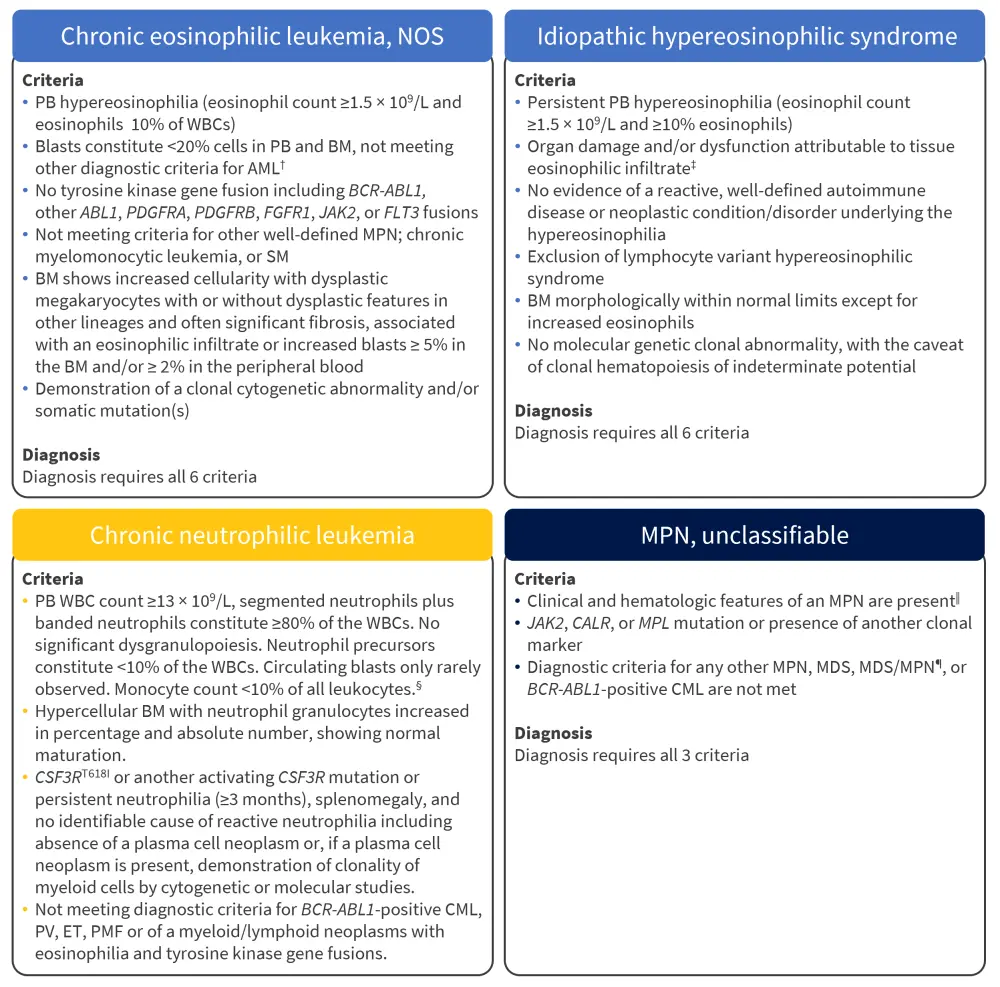
AML, acute myeloid leukemia; BM, bone marrow; CEL, chronic eosinophilic leukemia; CML, chronic myeloid leukemia; CNL, chronic neutrophilic leukemia; ET, essential thrombocythemia; iHES, idiopathic hypereosinophilic syndromes; MDS, myelodysplastic syndrome; MF, myelofibrosis; MPN, myeloproliferative neoplasm; MPN-U, myeloproliferative neoplasm unclassified; NOS, not otherwise specified; PB, peripheral blood; PMF, primary myelofibrosis; PV, polycythemia vera; SM, systemic mastocytosis; WBC, white blood cell.
*Adapted from Arber, et al.1
†AML with recurrent genetic abnormalities with <20% blasts is excluded.
‡Hypereosinophilia of uncertain significance has no tissue damage but fulfills the same diagnostic criteria.
§10–19% blasts in PB or BM represent CNL in accelerated phase; ≥20% blasts represents blast phase.
‖In cases presenting with BM fibrosis reactive causes must be excluded, including BM fibrosis secondary to infection, autoimmune disorder or other chronic inflammatory conditions, hairy cell leukemia or another lymphoid neoplasm, metastatic malignancy, or toxic (chronic) myelopathy.
¶In cases presenting with myelodysplastic features effects of any previous treatment, comorbidity, and changes during the progression of the disease process must be carefully excluded.
Review of the ICC by Thiele, et al.2
A critical review of the new ICC by several experts, including MPN Hub Steering Committee members Alessandro Vannuchi and Tiziano Barbui, has been recently published in the American Journal of Hematology. This highlighted that BM morphology remains central to MPN diagnosis and classification, whilst genetic factors also contribute to the evaluation of clonality, subtype, and prognosis.
The group reflected on the prognostic impact of several mutations, such as JAK2V617F, CALR, and MPL, and noted that the absence of BCR-ABL1-specific disease markers has hampered diagnostic progress and the development of disease-modifying drugs. However, they concluded that the new ICC framework is universally suitable, providing sufficient direction for the diagnosis and allocation of MPN subtypes.
Conclusion
The new ICC presents an updated approach to clinical presentation, morphological features, and disease specific mutations, as outlined previously in the 2016/2017 WHO classification. With refinement of the diagnostic criteria, the hope is that clinicians will now be able to distinguish between specific subtypes with greater accuracy and present a more definitive diagnosis for patients.
References
Please indicate your level of agreement with the following statements:
The content was clear and easy to understand
The content addressed the learning objectives
The content was relevant to my practice
I will change my clinical practice as a result of this content

