All content on this site is intended for healthcare professionals only. By acknowledging this message and accessing the information on this website you are confirming that you are a healthcare professional. If you are a patient or carer, please visit the MPN Advocates Network.
The MPN Hub website uses a third-party service provided by Google that dynamically translates web content. Translations are machine generated, so may not be an exact or complete translation, and the MPN Hub cannot guarantee the accuracy of translated content. The MPN Hub and its employees will not be liable for any direct, indirect, or consequential damages (even if foreseeable) resulting from use of the Google Translate feature. For further support with Google Translate, visit Google Translate Help.
The MPN Hub is an independent medical education platform, sponsored by AOP Health, GSK, and Novartis, and supported through independent educational grants from Blueprint Medicines, Incyte, Bristol Myers Squibb and Merck Sharp & Dohme LLC, Rahway, NJ, USA. The funders are allowed no direct influence on our content. The levels of sponsorship listed are reflective of the amount of funding given. View funders.
Now you can support HCPs in making informed decisions for their patients
Your contribution helps us continuously deliver expertly curated content to HCPs worldwide. You will also have the opportunity to make a content suggestion for consideration and receive updates on the impact contributions are making to our content.
Find out more
Create an account to access:
Bookmark & personalize site content
Receive alerts for new content in your areas of interest
View MPN content recommended for you
MOMENTUM study of momelotinib for myelofibrosis meets primary endpoint
On January 25, 2022, it was announced that the phase III, pivotal MOMENTUM (NCT04173494) study, evaluating momelotinib for the treatment of patients with symptomatic myelofibrosis (MF), achieved both its primary and major secondary endpoints; momelotinib has been associated with statistically significant improvements in symptoms, spleen size, and anemia.
Momelotinib
Momelotinib is an orally available inhibitor of Janus kinase 1 and 2 (JAK1/2) and activin A receptor type I/activin receptor-like kinase-2 (ALK-2). The agent is currently being evaluated across a number of clinical trials for the treatment of patients with MF who have received prior treatment with a U.S. Food and Drug administration (FDA)-approved JAK inhibitor.
MOMENTUM
MOMENTUM is a phase III, double-blind, randomized study investigating momelotinib versus danazol in patients aged ≥18 years with MF who meet the following eligibility criteria:
- Symptomatic with a Total Symptom Score (TSS) of ≥10.
- Anemic with hemoglobin of <10 g/dL at baseline.
- Prior treatment with a FDA-approved JAK inhibitor.
Across 21 countries, 195 patients were enrolled and randomized 2:1 to momelotinib (n = 130) and danazol (n = 65) treatment arms. Patients receiving danazol were able to transfer to momelotinib after 24 weeks of treatment, or earlier in the instance of splenic progression.
The study used the following endpoints to establish the value of momelotinib in MF:
- Primary endpoint: ≥50% reduction of TSS at the end of Week 24 compared to baseline.
- Secondary endpoints: transfusion independence for ≥12 weeks with maintained hemoglobin at ≥8 g/dL, and splenic response rate of ≥35% volume reduction at Week 24.
Results
Topline data from the MOMENTUM study demonstrate the encouraging efficacy and safety profiles of momelotinib versus danazol for patients with MF (Figure 1).
Figure 1. A Efficacy and B safety outcomes observed in patients enrolled in the MOMENTUM study*
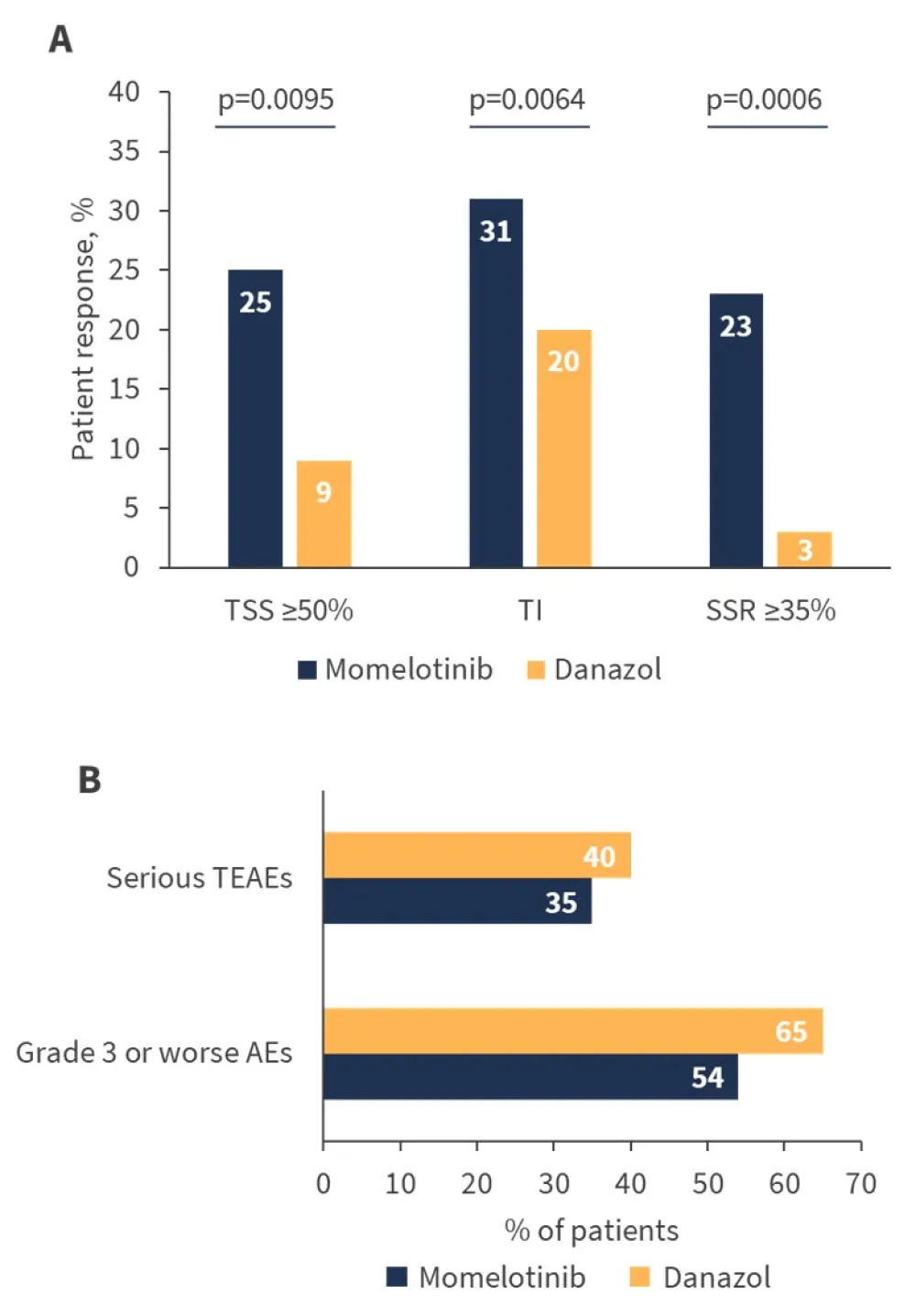
AEs, adverse events; SRR, splenic response rate; TEAEs, treatment-emergent adverse events; TI, transfusion independence; TSS, Total Symptom Score.
*Data from BusinessWire1
Findings from the MOMENTUM study suggest that momelotinib represents a promising treatment approach to MF with anemia, or patients with MF who may progress to symptomatic anemia.
References
Please indicate your level of agreement with the following statements:
The content was clear and easy to understand
The content addressed the learning objectives
The content was relevant to my practice
I will change my clinical practice as a result of this content

