All content on this site is intended for healthcare professionals only. By acknowledging this message and accessing the information on this website you are confirming that you are a healthcare professional. If you are a patient or carer, please visit the MPN Advocates Network.
The MPN Hub website uses a third-party service provided by Google that dynamically translates web content. Translations are machine generated, so may not be an exact or complete translation, and the MPN Hub cannot guarantee the accuracy of translated content. The MPN Hub and its employees will not be liable for any direct, indirect, or consequential damages (even if foreseeable) resulting from use of the Google Translate feature. For further support with Google Translate, visit Google Translate Help.
The MPN Hub is an independent medical education platform, sponsored by AOP Health, GSK, and Novartis, and supported through educational grants from Blueprint Medicines, Incyte, and Bristol Myers Squibb. The funders are allowed no direct influence on our content. The levels of sponsorship listed are reflective of the amount of funding given. View funders.
Now you can support HCPs in making informed decisions for their patients
Your contribution helps us continuously deliver expertly curated content to HCPs worldwide. You will also have the opportunity to make a content suggestion for consideration and receive updates on the impact contributions are making to our content.
Find out more
Create an account and access these new features:
Bookmark content to read later
Select your specific areas of interest
View MPN content recommended for you
ASH 2016 | Interim PET-Based Outcome Prediction and Treatment Changes in Patients with T-Cell Lymphomas Participating in the PETAL Trial
The 58th Annual Meeting & Exposition of the American Society of Hematology’s (ASH) took place in San Diego, CA, and on December 3rd, Andreas Hüttmann, MD, from University Hospital Essen, Germany, presented analysis of peripheral T-cell lymphoma patients recruited to the PETAL trial. The PETAL trial concluded that while treatment changes based upon interim-PET had no impact on outcome, it was reliable at predicting OS and time to treatment failure (TTTF).
From 1072 patients recruited to the PETAL trial, there were 76 patients with T-cell lymphomas of which 21 were ALK positive ALCL, 13 ALK negative ALCL, 18 with AITL, and 20 unspecified PTCL. Treatment regimen is described in the abstract, but briefly was 2 cycles of R-CHOP after a positive baseline PET result, CD20 patients did not receive rituximab but did receive four extra cycles of CHOP following favorable iPET (defined as Part A/favorable iPET). Different treatment arms were defined after iPET with unfavorable iPET response treatment being either 6 more cycles of CHOP or a complex 6 five day chemotherapy cycles with a complex of cytarabine, methotrexate, and etoposide treatment schedule (Part B/iPET unfavorable).
Highlights:
- iPET favorable in 75% of PTCL pts.
- iPET was shown to be predictive of outcome
- iPET favorable had significantly higher OS and TTTF than iPET unfavorable
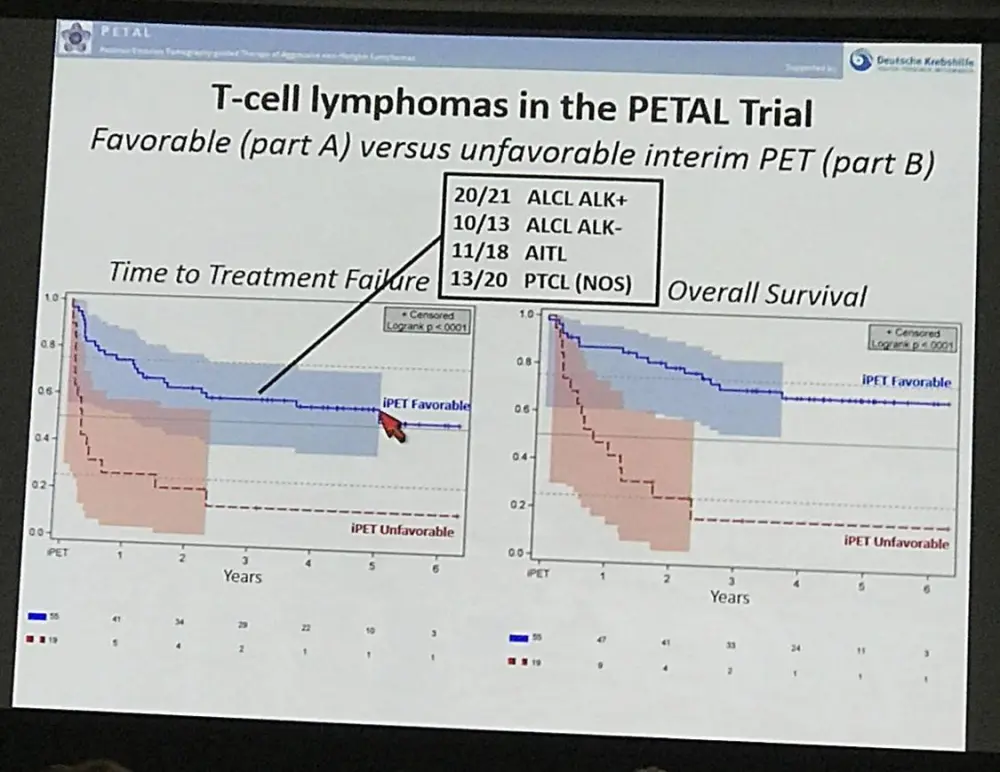
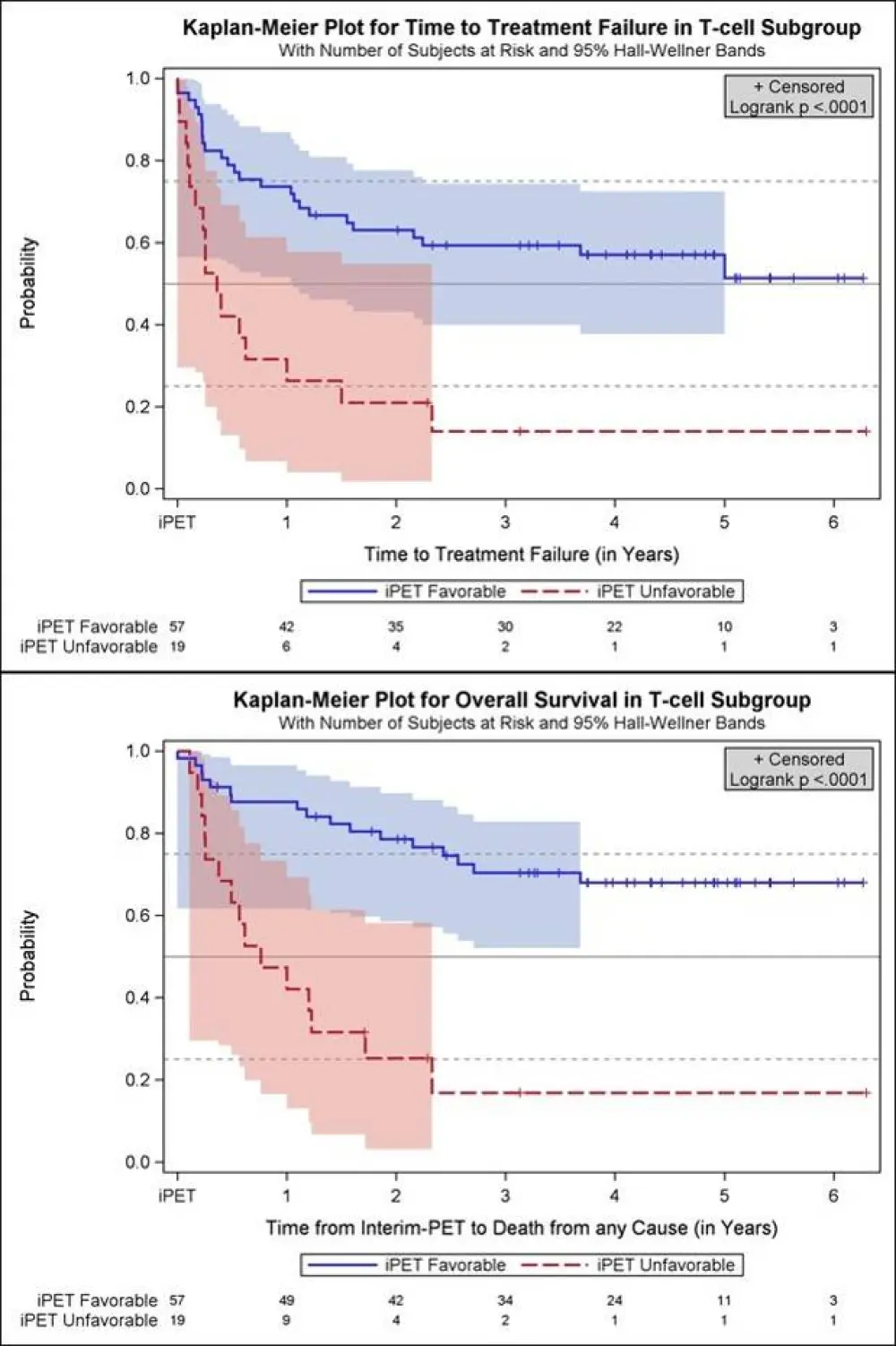
- 2 year probability TTTF: iPET favorable = 63%, iPET unfavorable = 21% (P<0.0001)
- 2 year probability OS: iPET favorable = 79%, iPET unfavorable = 25% (P<0.0001)
- Following unfavorable iPET, change from CHOP to other treatment did not improve OS or TTTF
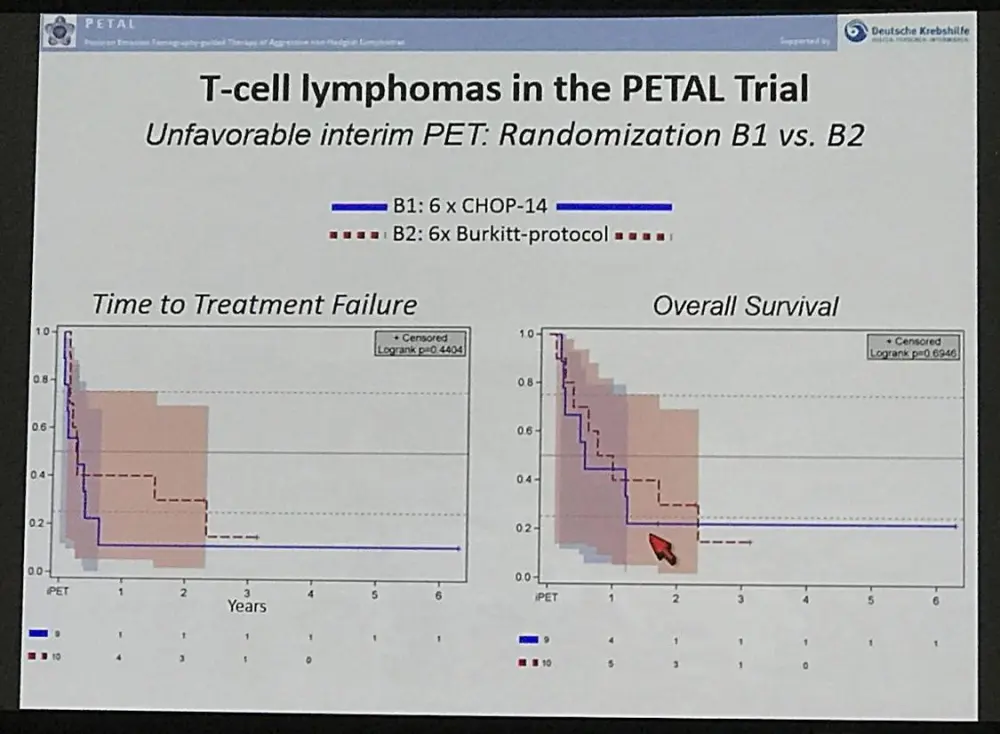
- Part B treatment regimen produced much higher AEs than CHOP after unfavorable iPET

Andreas Hüttmann concluded by stating that iPET was highly predictive of PTCL patient outcome, and that following an unfavorable iPET switching to a more aggressive treatment schedule could not improve outcome. Other approaches will need to be brought forward in order to improve outcome in PTCL patients who do not respond to initial CHOP treatment.

Abstract:
Introduction: The PETAL trial is a multicenter randomized controlled study for patients with aggressive lymphomas of diverse histologies (EudraCT 2006-001641-33, NCT00554164). In the study population as a whole interim PET (iPET) reliably predicted time to treatment failure (TTTF) and overall survival (OS). Interim PET-based treatment changes, however, had no impact on outcome (ASH 2014, abstract 391). Here we report the exploratory analysis for peripheral T cell lymphomas (PTCL).
Methods: Pts. aged 18 to 80 yrs. with newly diagnosed aggressive lymphomas and a positive baseline PET received 2 cycles of rituximab (R), cyclophosphamide, doxorubicin, vincristine and prednisone (CHOP) followed by iPET. R was omitted in pts. with CD20-negative lymphomas. The conditions of iPET were strictly defined: 3-week interval between the 2nd (R-)CHOP cycle and iPET to avoid inflammatory reactions (Eur J Nucl Med Mol Imaging 30:682, 2003), no G-CSF after the 2nd cycle to avoid altered glucose biodistribution (J Nucl Med 47:950, 2006), standardized uptake value (SUV)-based PET interpretation to improve reproducibility (favorable iPET response: reduction of maximum SUV by > 66 % compared to baseline; J Nucl Med 48:1626, 2007). PTCL pts. with CD20-negative lymphomas and a favorable iPET uniformly received 4 additional cycles of CHOP (part A of the trial). Pts. with an unfavorable iPET were randomized to continue CHOP for 6 additional cycles or receive 6 blocks of a more complex methotrexate-, cytarabine- and etoposide-based regimen originally designed for Burkitt lymphoma (Blood 124: 3870, 2014; part B). Sample size of the entire study population was based on the empirically derived assumption that treatment failure after 2 yrs. (progression, relapse, treatment discontinuation due to toxicity, start of alternative therapy, death of any cause) could be improved from 30 % to 45 % in part B (alpha=0.05, power=0.8). Secondary endpoints included OS and toxicity.
Results: Fifty-seven oncological centers and 23 nuclear medicine institutions participated in the trial. Between 2007 and 2012 1072 pts. were registered, and 862 (80.4 %) had a positive baseline PET, received 2 cycles (R-)CHOP, underwent iPET and were allocated to one of the post-iPET treatment arms detailed above. Reference pathology was available in 98 %, and median follow-up is 52 months. All in all, there were 76 pts. (8.8 % of all treated pts.) with T-cell lymphomas of whom 21 had ALK+ anaplastic large cell lymphoma (ALCL), 13 ALK- ALCL, 18 angioimmunoblastic T-cell lymphoma (AITL) and 20 PTCL not otherwise specified (NOS). Interim PET was favorable in 57 pts. (75 %) and unfavorable in 19 pts. with T-cell lymphomas (25 %). It was highly predictive of outcome, TTTF and OS being significantly higher in part A than B (2-year probability for TTTF: 63 % vs. 21 %; univariate hazard ratio (HR) for B 3.4, 95 % confidence interval (CI) 1.8 - 6.4, p<0.0001; OS: 79 % vs. 25 %; univariate hazard ratio (HR) for B 5.0, 95 % CI 2.4 - 10.3, p<0.0001; Figure). Interestingly, the proportion of T-cell lymphoma pts. with an unfavorable iPET response was more than twice as high as the corresponding proportion of B-cell lymphoma pts., and the difference in survival between pts. with a favorable vs. unfavorable iPET response was more pronounced in T-cell lymphomas than in B-cell lymphomas.
TTTF (2-year probability: 81 % vs. 46 % vs. 49 % vs. 35 %; p=0.0110) and OS (90 % vs. 69 % vs. 52 % vs. 50 %; p=0.0026) were better in ALK+ ALCL than in ALK- ALCL, AITL or PTCL NOS. In pts. with an unfavorable iPET response, a switch from CHOP to the alternative regimen failed to improve TTTF or OS. The latter was associated with more frequent grade 3/4 neutropenia (40 % vs. 0 % vs. 11 %, p=0.0279), thrombocytopenia (70 % vs. 33 % vs. 23 %; p=0.0106), infection (60 % vs. 44 % vs. 18 %, p=0.0057) and mucositis (40 % vs. 33 % vs. 4 %, p=0.0025) as compared to 6 or 4 post-iPET cycles of CHOP, respectively, but treatment-related mortality was similar in all treatment arms (2 vs. 1 vs. 2 deaths).
Conclusion: In this large multicenter trial iPET proved highly predictive of outcome in PTCL. A favorable iPET was found in 75 % of pts., and this was associated with long-term survival in about 70 %. In pts. with an unfavorable iPET response, outcome was dismal and could not be improved by switching to a more aggressive regimen. Novel strategies are required for PTCL pts. failing to respond to the first 2 cycles of CHOP.
References
Please indicate your level of agreement with the following statements:
The content was clear and easy to understand
The content addressed the learning objectives
The content was relevant to my practice
I will change my clinical practice as a result of this content
Your opinion matters
What is the primary reason you use bridging therapy in patients with DLBCL awaiting CAR T-cell therapy?

