All content on this site is intended for healthcare professionals only. By acknowledging this message and accessing the information on this website you are confirming that you are a healthcare professional. If you are a patient or carer, please visit the MPN Advocates Network.
The MPN Hub website uses a third-party service provided by Google that dynamically translates web content. Translations are machine generated, so may not be an exact or complete translation, and the MPN Hub cannot guarantee the accuracy of translated content. The MPN Hub and its employees will not be liable for any direct, indirect, or consequential damages (even if foreseeable) resulting from use of the Google Translate feature. For further support with Google Translate, visit Google Translate Help.
The MPN Hub is an independent medical education platform, sponsored by AOP Health, GSK, and Novartis, and supported through educational grants from Blueprint Medicines, Incyte, and Bristol Myers Squibb. The funders are allowed no direct influence on our content. The levels of sponsorship listed are reflective of the amount of funding given. View funders.
Now you can support HCPs in making informed decisions for their patients
Your contribution helps us continuously deliver expertly curated content to HCPs worldwide. You will also have the opportunity to make a content suggestion for consideration and receive updates on the impact contributions are making to our content.
Find out more
Create an account and access these new features:
Bookmark content to read later
Select your specific areas of interest
View MPN content recommended for you
Splanchnic vein thrombosis: Impact on survival outcomes in younger patients with PV or ET
Splanchnic vein thrombosis (SVT) occurs in approximately 5% of patients with myeloproliferative neoplasms (MPN) and development is commonly associated with the Janus kinase 2V617F driver mutation either at diagnosis or follow-up.1
Here, we summarize a study published by Garrote et al.1 in Annals of Hematology on the role of SVT and genomic characteristics in the outcomes and survival of patients with polycythemia vera and essential thrombocythemia.
Patient population1
- A cohort of 255 patients aged 37–60 years were included:
- 90 patients with SVT, with 59 of those presenting at MPN diagnosis, 10 before diagnosis, and 21 during follow-up.
- 165 patients with MPN but without SVT as a control group
- Localization of SVT:
- Splenoportal area, 64%
- Hepatic vein (Budd-Chiari syndrome), 32%
- Both locations, 3%
- The incidence of heterozygous Janus kinase 2 mutation was more common in patients with SVT compared with the control group (87% vs 69%, respectively; p < 0.05).
Key findings1
- Compared with the control cohort, patients with SVT presented with a higher risk of:
- Venous rethrombosis; hazard ratio (HR) 3.0 (95% confidence interval [CI], 1.5–6.0; p = 0.003) and after correction by age; HR 1.1, (95% CI, 1.08–1.15; p < 0.001)
- Total thrombosis; HR 3.2, (95% CI, 1.6–6.4; p = 0.001) and after correction by age; HR 1.03 (95% CI, 0.99–1.06; p = 0.07)
- Thrombosis at 10 years was 27% and 8%, respectively (p = 0.001).
- Patients with SVT also presented with:
- lower rates of event-free survival; HR 3.0 (95% CI, 1.9–4.8; p < 0.001); and
- association between molecular high-risk status and an increased risk of venous rethrombosis; HR 5.8 (95% CI, 1.4–24.0, p = 0.01).
- Patients who developed SVT during the follow-up period were more likely to be molecular high-risk than those with SVT present at diagnosis (52% vs 13%, respectively; p < 0.05).
- Median overall- and event-free survival was poorer in patients with SVT (Figure 1)
Figure 1. Median overall- and event-free survival in patients with or without SVT*
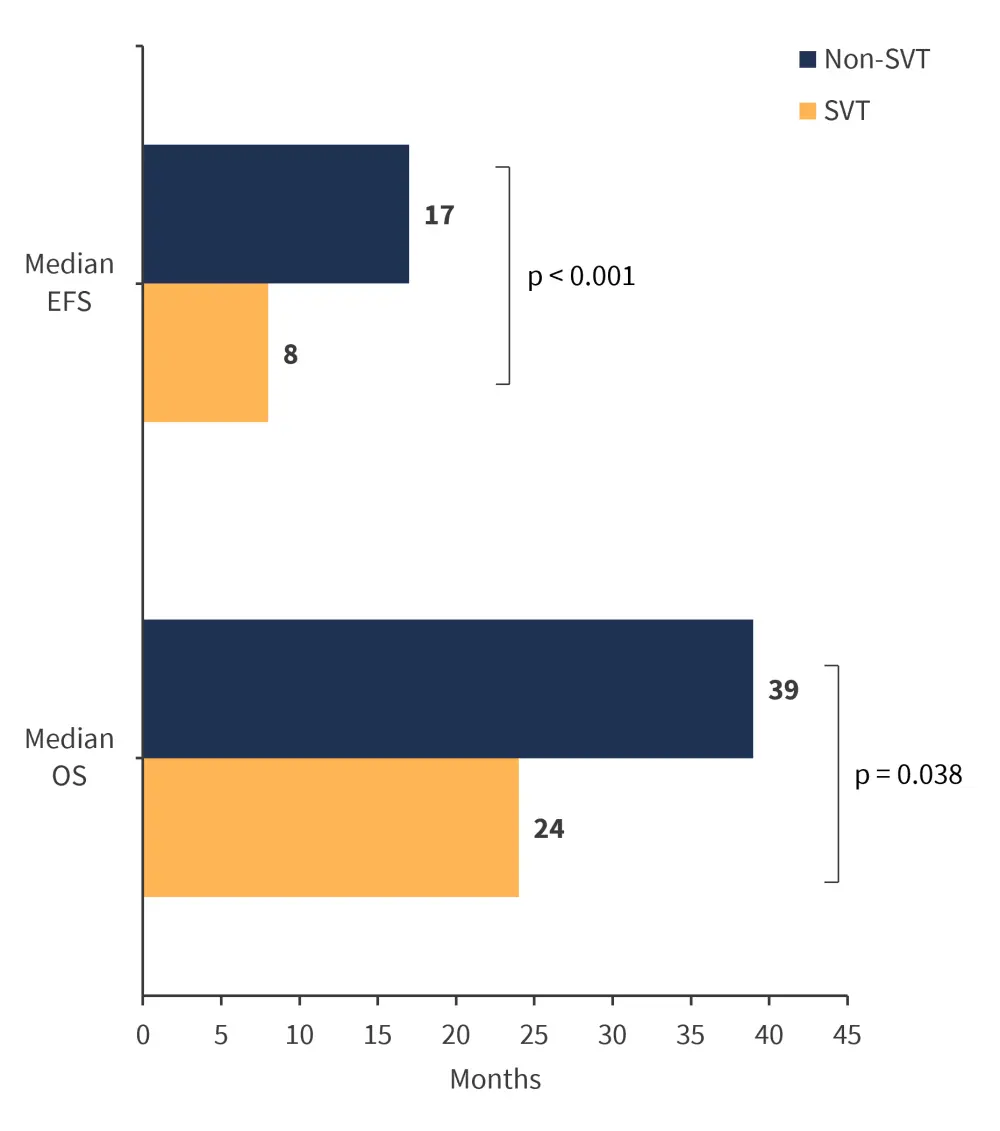
EFS, event-free survival; OS, overall survival; SVT, splanchnic vein thrombosis.
*Data from Garrote, et al.1
|
Key learnings |
|---|
|
References
Please indicate your level of agreement with the following statements:
The content was clear and easy to understand
The content addressed the learning objectives
The content was relevant to my practice
I will change my clinical practice as a result of this content



